- Nanocrystalline materials are microscopic crystals too small to see with the naked eye
- Customizability makes them ideal candidates for strong and lightweight structural materials
- High performance computing makes discovering new materials more efficient
Have you ever stepped out into a clear night and marveled at how small you are? With alone, it’s easy to get lost in the enormity of the cosmos. But the other side of the size scale is equally incredible.
 Just as humans are minuscule in the face of the galaxy, we are giants compared to —tiny substances that could have a huge impact on our future.
Just as humans are minuscule in the face of the galaxy, we are giants compared to —tiny substances that could have a huge impact on our future.
Defined by a grain size of less than 100 nanometers – for scale, a single red blood cell is around – these materials fascinate materials scientist Dr. Kris Darling and professor .
“We are looking for a material that is strong, light, and able to withstand multitudes of different external stimuli. They behave very differently compared to traditional material,” says Solanki.
Nanocrystalline structures have been investigated for the past thirty years, but so far no material has proven well-suited for structural applications, due to microstructural instability in the face of external stimuli such as temperature, radiation, magnetic fields, etc.
“Our interest is to make these materials more viable for advanced application,” Solanki says. “So then we can have an impact on sustainable development.” If he succeeds, it could transform areas as diverse as transportation, space travel, and energy production.
Building a better tomorrow
In essence, Darling, Solanki, and their team are trying to find materials that have a variety of beneficial properties. Increased efficiency is the overall goal they’re trying to achieve.
 “An aircraft’s speed at 30,000 feet is about 500 miles per hour,” says Darling. “How do we increase that by maybe more than 100 miles an hour? You need stronger alloys and materials that are durable enough to be able to travel that far. This allows you to use less fuel and travel faster. If efficiency is higher, you're going to reduce your carbon footprint.”
“An aircraft’s speed at 30,000 feet is about 500 miles per hour,” says Darling. “How do we increase that by maybe more than 100 miles an hour? You need stronger alloys and materials that are durable enough to be able to travel that far. This allows you to use less fuel and travel faster. If efficiency is higher, you're going to reduce your carbon footprint.”
While properties like “resilient” and “stronger” sound promising, it’s not like Darling and Solanki can just ask the materials to perform how they want them to. Instead, they have to find the right material for the right job. To do this, they rely on high-performance computing (HPC) to help identify the most promising materials to test.
In this case, that’s the AGAVE cluster at ASU. With 8,512 cores on 324 hyper-converged, multi-processor X86 Intel Xeon E5-2680v4 “Broadwell” 2.4GHz processors and 1,280 cores on Intel Xeon Phi 7210 “Knights Landing” processors, AGAVE has the computing power to produce results more quickly.
“We use the computer for a ‘materials by design approach,’ where we use computational tools to identify the weakest links or weakest behavior that we can control,” says Solanki. “Then with the help of ARL, we fine-tune to make sure the materials that we're going to develop will behave a certain way.”
Stepping into the real world
Eventually, these materials need to get out of the computer and into the lab. After settling on a promising nanomaterial recipe, Darling places copper and powders in an industrial shaker.
 “We have milling balls inside the shaker which makes the particles smaller and finer, so that they mix properly together,” says Darling. “You have different size balls inside the blender and you shake them very vigorously at liquid nitrogen temperature, because you want to cool the system down.”
“We have milling balls inside the shaker which makes the particles smaller and finer, so that they mix properly together,” says Darling. “You have different size balls inside the blender and you shake them very vigorously at liquid nitrogen temperature, because you want to cool the system down.”
He continues, “And then it comes out as a powder, which is passed through a press and consolidated at high temperature into a rod. And then we use the rod for testing at ASU.”
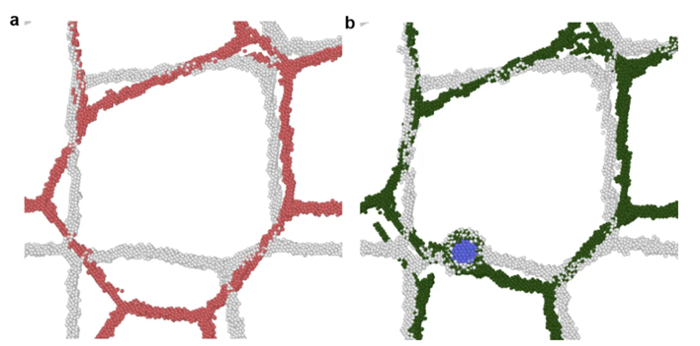
But discovery is not a single step process. Once the material is fabricated, the team returns to computation to help them understand what really happened in the lab.
“The computation tells you what will happen, but then you perform the experiment and something unexpected happens,” says Solanki. “So then you go back to the computation and it tells you why that happened. It’s a closed loop that allows you to iterate and fine-tune the material.”
While many labs are experimenting with nanocrystalline materials, Darling and Solanki believe their team’s approach is unique. But even so, they would like to see greater collaboration within the field.
“One of the things that is missing is the community coming together and realizing the potential of nanocrystalline materials and making an effort towards developing it. This is the key to developing many more new applications and alloys that were not feasible before.”
Solanki continues, “Looking back at the amount of work we have accomplished in just four years, we think that in ten years from now we will have a greater impact than you can imagine. When we consider that the work we’re doing could change many things and could impact life—that motivates us every single day.”
Read more:
